|
These books contain exercises and tutorials to improve your practical skills, at all levels.This site is a Google powered search engine that queries Google to show PDF search results.Please respect the publisher and the author for their creations if their books are copyrighted.Please contact us or the content providers to delete copyright contents if any and email us, well remove relevant links or contents immediately.
Submit us a DMCA notice and Inform about office files copyright abuse, using contact form. This site does not host pdf files all document are the property of their respective owners. All eBooks displayed on this site may be used for educational purposes only. When effervescence fizzingbubbling out of the gas is over, excess of the insoluble carbonate is filtered off. 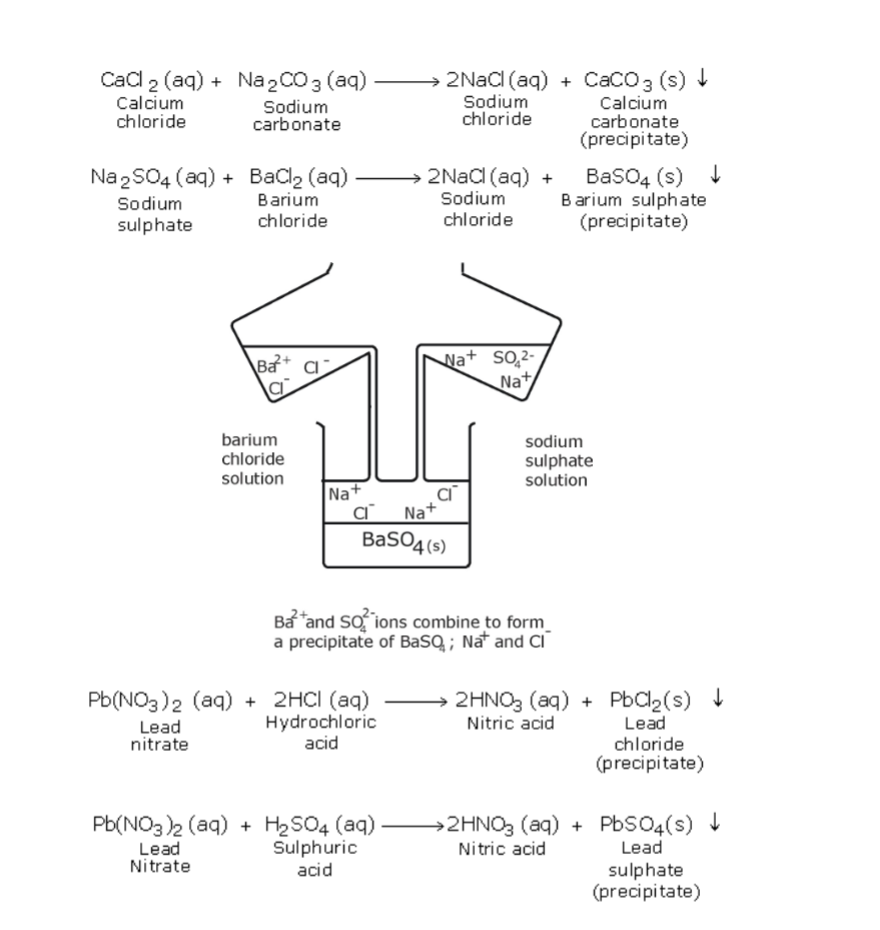
Table of Some Complex Salts Table of Some Double Salts Table of Solubility of Salts Preparation of Salts Soluble Salts Insoluble Salts Effect of Heat on Salts Effect of Heat on Chlorides Effect of Heat on Nitrate(V) Effect of Heat on Sulphate(VI) Effect of Heat on Carbonates(IV) and Hydrogen Carbonate(IV). Introduction to Salts A salt is an ionic compound formed when the cation from a base combine with the anion derived from an acid. A salt is therefore formed when the hydrogen ions in an acid are replaced whollyfully or partiallypartly,directly or indirectly by a metal or ammonium radical. The number of ionizablereplaceable hydrogen in an acid is called basicity of an acid. Some acids are therefore: monobasic acids generally denoted H X e.g. H Cl, H NO 3,HCOO H,CH 3 COO H. A normal salt is formed when all the ionizable replaceable hydrogen in an acid is replaced by a metal or metallic ammonium radical. An acid salt is formed when partportion the ionizable replaceable hydrogen in an acid is replaced by a metal or metallic ammonium radical. Hygroscopic salts compounds are those that absorb water from the atmosphere but do not form a solution. Some salts which are hygroscopic include anhydrous copper(II)sulphate(VI), anhydrous cobalt(II)chloride, potassium nitrate(V), common table salt. Deliquescent salts compounds are those that absorb water from the atmosphere and form a solution. 
Efflorescent saltscompounds are those that lose their water of crystallization to the atmosphere. Some salts which effloresces include: sodium carbonate decahydrate, Iron(II)sulphate(VI)heptahydrate, sodium sulphate (VI)decahydrate. Some salts contain water of crystallization.They are hydrated.Others do not contain water of crystallization. Table of Some Complex Salts Name of complex salt Chemical formula Colour of the complex salt Tetraamminecopper(II)sulphate(VI) Cu(NH 3 ) 4 SO 4 H 2 O Royaldeep blue solution Tetraamminezinc(II)nitrate(V) Zn(NH 3 ) 4 (NO 3 ) 2 Colourless solution Tetraamminecopper(II) nitrate(V) Cu(NH 3 ) 4 (NO 3 ) 2 Royaldeep blue solution Tetraamminezinc(II)sulphate(VI) Zn(NH 3 ) 4 SO 4 Colourless solution Some salts exist as two salts in one. 
Soluble Salts Soluble salts may be prepared by using any of the following methods: (i) Direct displacementreaction of a metal with an acid. Chemistry Form 2 Series Than HydrogenBy reacting a metal higher in the reactivity series than hydrogen with a dilute acid, a salt is formed and hydrogen gas is evolved. Excess of the metal must be used to ensure all the acid has reacted. When effervescencebubbling fizzing has stopped,excess metal is filtered. The filtrate is heated to concentrate then allowed to crystallize. The filtrate is heated to concentrate, allowed to crystallize then washed with distilled water before drying between filter papers. By adding an excess of a soluble insoluble carbonate or hydrogen carbonate to adilute acid, effervescence fizzingbubbling out of carbon(IV)oxide gas shows the reaction is taking place.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Home
- About
- Donate
- Blog
- Bitrix24 app
- Reason definition
- How to use disk genius repair bad hard drive
- Midnight club 3 dub edition remix xbox cheats
- Matlab 2009 free download for windows 7 32 bit
- Black ops 2 gsc studio private match
- Rix lounge
- Chem c3000 store
- Microsoft word cost
- Garritan jazz and big band 3 crack
- Webscraper out of selenium
- Amped five software free trial
- Amadeus full movie
- Gta romania 2 telecharger
 RSS Feed
RSS Feed
